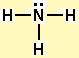NH3 - Ammonia:
First draw the Lewis dot structure:
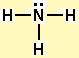
Electron geometry: tetrahedral. Hybridization: sp3
Then draw the 3D molecular structure using VSEPR rules:
The molecular geometry of NH3 is trigonal pyramidal with asymmetric charge distribution on the central atom.
Therefore this molecule is polar.
Ammonia on Wikipedia.
Back to Molecular Geometries & Polarity Tutorial: Molecular Geometry & Polarity Tutorial.
For homework help in math, chemistry, and physics: www.tutor-homework.com.